45 isotopes and average atomic mass worksheet
1. Complete the chart for the elements and isotopes below Average Atomic Mass. Name Key. Be sure to show all of your work. 85. 1. Rubidium is a soft, silvery-white metal that has two common isotopes, Rb and. Join LiveJournal Password requirements: 6 to 30 characters long; ASCII characters only (characters found on a standard US keyboard); must contain at least 4 different symbols;
Isotopes Average Atomic Mass Oct 27, 2019 ... Isotopes and Average Atomic Mass. Isotopes are atoms of the same element (they have the same number of protons) but with different.
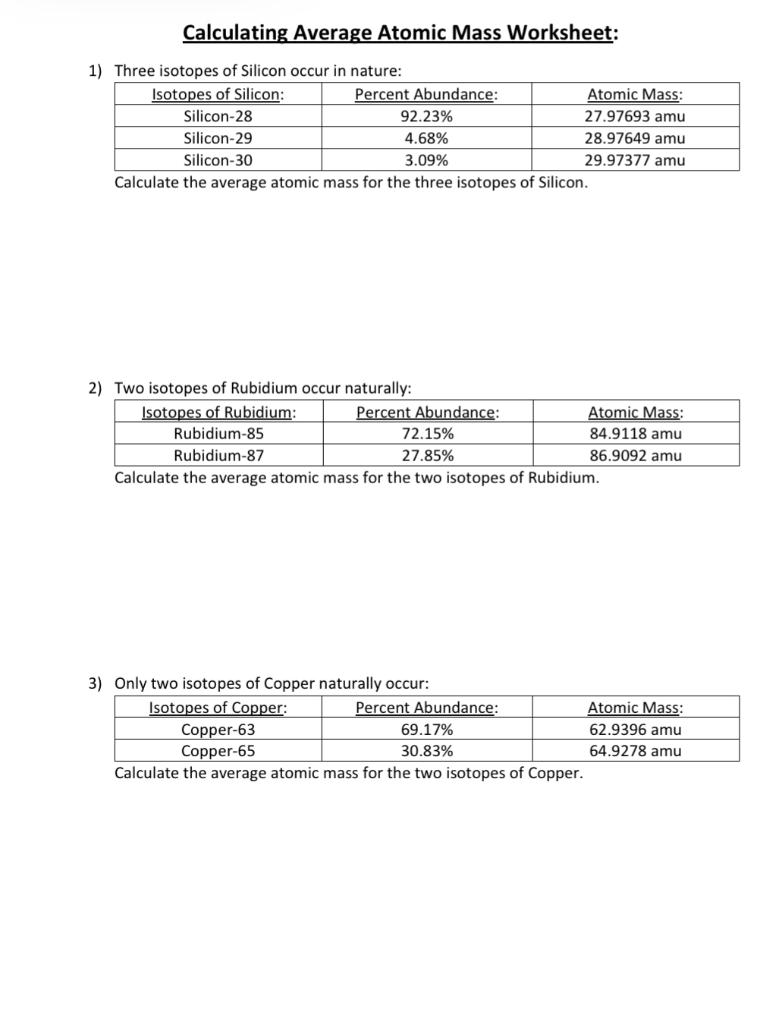
Isotopes and average atomic mass worksheet
Average Atomic Mass Practice Problems - scott.kyschools.us 1. What is the atomic mass of hafnium if, out of every 100 atoms, 5 have a mass of 176, 19 have a mass of 177, 27 have a mass of 178, 14 have a mass of 179, and 35 have a mass of 180.0? 2. Calculate the average atomic mass of lithium, which occurs as two isotopes that have the following atomic masses and abundances in nature: 6.017 u, 7.30% and Identifying Elements and Masses Using a Mass Spectrum of an Element … Vanadium has an average atomic mass of 50.94 amu and manganese has an average atomic mass of 54.94 amu. Neither of the other masses would make sense with a major isotope of 52 amu. This element is ... How to Calculate Average Atomic Mass | Chemistry | Study.com How to Calculate Average Atomic Mass: Example 1. Calculate the average atomic mass of boron given that 19.8% of its naturally occurring atoms have a mass of 10.013 amu and 80.2% have a mass of 11. ...
Isotopes and average atomic mass worksheet. Atom and Isotope WS_ANS KEY - WongChemistry These atoms differ in the number of neutrons. The average atomic mass is the weighted average of all the isotopes of an element. Example: A sample of cesium ... Atomic Mass Worksheet Average Atomic Mass Worksheet: show all work. 1) Rubidium is a soft, silvery-white metal that has two common isotopes," Rb and Rb. If. 2.3: Calculating Atomic Masses (Problems) - Chemistry LibreTexts Average atomic masses listed by IUPAC are based on a study of experimental results. Bromine has two isotopes, 79 Br and 81 Br, whose masses (78.9183 and 80.9163 amu) and abundances (50.69% and 49.31%) were determined in earlier experiments. Calculate the average atomic mass of Br based on these experiments. Atomic Structure Worksheet - Washoe County School District The atomic number gives the “identity “of an element as well as its location on the Periodic Table. No two different elements will have the atomic number. The of an element is the average mass of an element’s naturally occurring atoms, or isotopes, taking into account the of each isotope.
Unit 3: Atomic Theory & Structure The isotope with a mass of. 11.009 amu (11X) has a relative abundance of. 80.09%. Calculate the atomic mass of this element. (10.012) (0.1991) = 1.993. (11.009)( ... Quiz & Worksheet - Atomic Number and Mass Number | Study.com Print Atomic Number and Mass Number Worksheet 1. If Atom #1 has 19 protons and 22 neutrons, and Atom #2 has 20 protons and 22 neutrons, are these isotopes of the same element? NAME Average Atomic Mass Worksheet: show all work. Calculate the average atomic mass. 35.46 amu 6) Copper used in electric wires comes in two flavors (isotopes): 63Cu and 65Cu. 63Cu has an atomic mass of 62.9298 amu and an abundance of 69.09%. The other isotope, 65Cu, has an abundance of 30.91%. The average atomic mass between these two isotopes is 63.546 amu. Calculate the actual atomic mass ... Isotope Aug 8, 2016 ... Worksheet - Isotopes and Average Atomic masses. Name. Period. Date. Key. 1. Four isotopes of lead include lead-204, lead-206, lead-207, ...
Isotopes and Atomic Mass - PhET Isotopes and Atomic Mass - PhET Quiz & Worksheet - Atomic Number and Mass Number Print Atomic Number and Mass Number Worksheet 1. If Atom #1 has 19 protons and 22 neutrons, and Atom #2 has 20 protons and 22 neutrons, are these isotopes of the same element? No, because they ... NAME Average Atomic Mass Worksheet: show all work. and 24.47 percent 37Cl (mass = 36.966 amu). Calculate the average atomic mass. 6) Copper used in electric wires comes in two flavors (isotopes): 63Cu and 65Cu. 63Cu has an atomic mass of 62.9298 amu and an abundance of 69.09%. The other isotope, 65Cu, has an abundance of 30.91%. The average atomic mass between these two isotopes is 63.546 amu. Isotopes-and-Average-Atomic-Mass-worksheet_Key-1.pdf Example: mass number. 15N+3 chorge. 7 protons. 8 neutrons (15-7). ISOTOPES AND AVERAGE. ATOMIC MASS. Name. Elements come in a variety of isotopes, ...
Build an Atom - Atoms | Atomic Structure | Isotope Symbols Build an atom out of protons, neutrons, and electrons, and see how the element, charge, and mass change. Then play a game to test your ideas! Then play a game to test your ideas! Skip to Main Content
Chemistry Honors Atomic structure Unit (Ch. 4) Atomic mass worksheet ... Bromine has two isotopes with the first having a mass of 78.918336 amu and ... (a) Calculate the atomic mass of europium.
Home | ExploreLearning Solve the math fact fluency problem. Adaptive and individualized, Reflex is the most effective and fun system for mastering basic facts in addition, subtraction, multiplication and division for grades 2+.
Atomic Structure Worksheet - Washoe County School District Theof an element is the average mass of an element 's naturally occurring atoms, or isotopes, taking into account theof each isotope. In order to calculate the number of neutrons you must subtract the from the. Give the symbol and number of protons in one atom of: Lithium Mercury Iron. Com lete the table below. Symbol Atomic Number Mass Number Number of Protons …
Success Essays - Assisting students with assignments online We offer the lowest prices per page in the industry, with an average of $7 per page. Success Essays Features. Get All The Features For Free. $11. per page. FREE Plagiarism report. FREE The best writer. FREE Formatting. FREE Title page. FREE Outline. Place An Order. We Offer the Custom Writing Service with 3 Key Benefits . Quality Essay Help. Our team of professional …
Calculating Average Atomic Mass Worksheet - Peoria Public Schools The element copper has naturally occurring isotopes with mass numbers of 63 and 65. The relative abundance and atomic masses are 69.2% for a mass of 62.93amu ...
Worksheet #4 – Isotopes & Average Atomic Mass Worksheet #1 – Calculations with Subatomic Particles ... Answer the following questions regarding average atomic mass and isotopes.
Lifestyle | Daily Life | News | The Sydney Morning Herald The latest Lifestyle | Daily Life news, tips, opinion and advice from The Sydney Morning Herald covering life and relationships, beauty, fashion, health & wellbeing
Cambridge IGCSE Chemistry Coursebook (fourth edition) - Issuu 09/06/2014 · Key definition relative atomic mass (Ar) – the average mass of normally occurring atoms of an element on a scale where the carbon-12 atom has a mass of exactly 12 units.
How to Calculate Average Atomic Mass | Chemistry | Study.com How to Calculate Average Atomic Mass: Example 1. Calculate the average atomic mass of boron given that 19.8% of its naturally occurring atoms have a mass of 10.013 amu and 80.2% have a mass of 11. ...
Identifying Elements and Masses Using a Mass Spectrum of an Element … Vanadium has an average atomic mass of 50.94 amu and manganese has an average atomic mass of 54.94 amu. Neither of the other masses would make sense with a major isotope of 52 amu. This element is ...
Average Atomic Mass Practice Problems - scott.kyschools.us 1. What is the atomic mass of hafnium if, out of every 100 atoms, 5 have a mass of 176, 19 have a mass of 177, 27 have a mass of 178, 14 have a mass of 179, and 35 have a mass of 180.0? 2. Calculate the average atomic mass of lithium, which occurs as two isotopes that have the following atomic masses and abundances in nature: 6.017 u, 7.30% and

0 Response to "45 isotopes and average atomic mass worksheet"
Post a Comment